Scientists present that lecanemab clears amyloid plaques by participating microglia by way of its Fc fragment, defining the mobile program behind its therapeutic impact
Lecanemab, marketed as Leqembi, is a monoclonal antibody remedy for Alzheimer’s disease that reduces the buildup of toxic amyloid plaques in the brain and slows cognitive decline. Scientists from VIB and KU Leuven have now uncovered exactly how it works.
Their study shows that a specific part of the antibody, known as the ‘Fc fragment,’ is crucial for activating microglia, the immune cells of the brain, and triggering the processes required to remove plaques. This provides the first clear mechanistic explanation for how this category of Alzheimer’s drugs achieves its effects.
The results, published in Nature Neuroscience, help resolve long-standing questions and may guide the development of safer and more effective therapies.
Fc fragment drives plaque clearance
“Our study is the first to clearly demonstrate how this anti-amyloid antibody therapy works in Alzheimer’s disease. We show that the therapy’s efficacy relies on the antibody’s Fc fragment, which activates microglia to effectively clear amyloid plaques,” says Dr. Giulia Albertini, co-first author of the study. “The Fc fragment works as an anchor that microglia latch onto when they are near plaques, as a consequence of which these cells are reprogrammed to clear plaques more efficiently.”
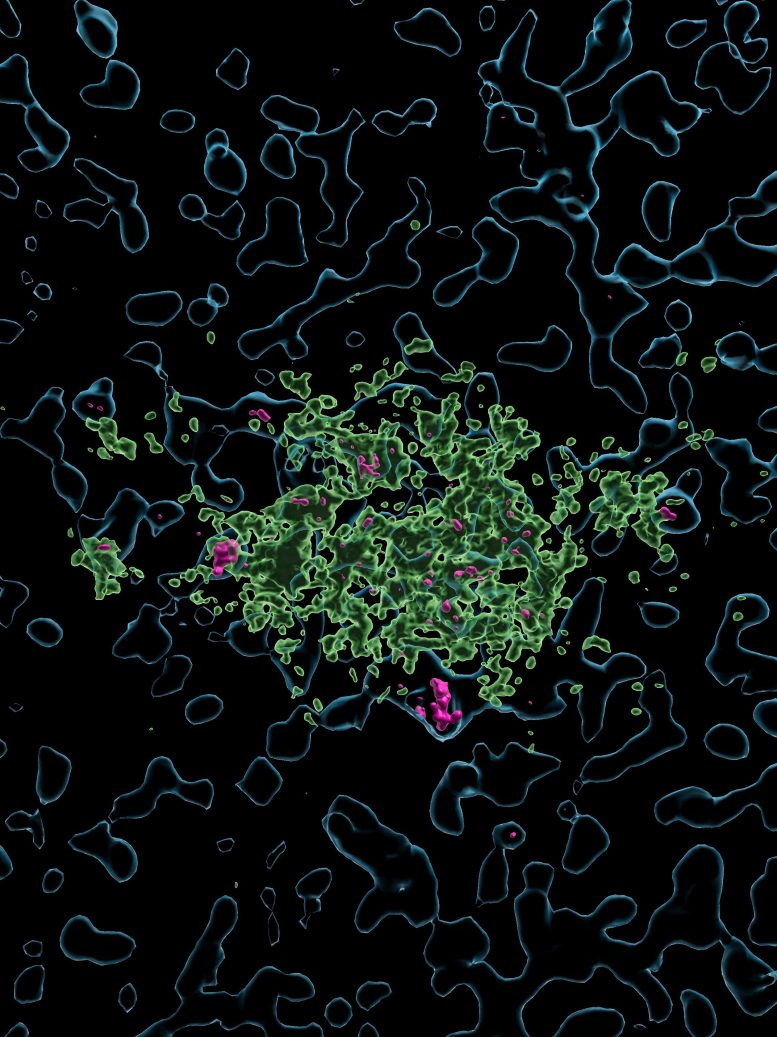
Alzheimer’s disease affects more than 55 million people worldwide. It is driven in part by the accumulation of amyloid plaques in the brain, clusters of misfolded proteins that damage neurons and eventually lead to dementia. Microglia normally gather around these plaques, but they are often unable to remove them effectively. In recent years, researchers have focused on therapies that aim to restore this natural cleanup function.
Antibody therapy restoring microglial function
Lecanemab is one of the first FDA-approved treatments designed to target amyloid-beta plaques and slow disease progression. Despite its clinical use, scientists have debated how exactly the antibody clears these toxic protein deposits, and concerns about side effects have complicated its broader application.
Antibodies are built from two main components. One region binds to a specific target, in this case, amyloid plaques. The other region, called the Fc fragment, interacts with the immune system. Earlier research suggested that activating microglia might be key to plaque removal, but there was no direct proof that this activity explained the therapeutic benefit of lecanemab.
Some researchers even proposed that plaques could be cleared without involvement of the Fc fragment. In the new study, the team led by Prof. Bart De Strooper demonstrated that the Fc fragment is indispensable. Microglia responded to the antibody only when this segment was intact and functional, confirming that immune activation through the Fc region is central to the drug’s effect.

Using an in-house Alzheimer’s mouse model engrafted with human microglial cells, the researchers created a controlled system in which they could examine how lecanemab activates human cells and how this leads to amyloid plaque clearance. The researchers also found that an antibody without the Fc fragment had no effect.
“The fact that we used human microglia within a controlled experimental model was a major strength of our study. This allowed us to test the very antibodies used in patients and observe human-specific responses with unprecedented resolution,” adds Magdalena Zielonka, co-first author.
The cellular processes of microglial clearance
The researchers then proceeded to elucidate how microglia clean the amyloid plaques in this hybrid human-animal model following lecanemab activation. They identified key cellular machinery needed to clear the amyloid plaques: namely, phagocytosis and lysosomal activity. Without the Fc fragment, none of these critical cellular processes were triggered. By combining single-cell and spatial transcriptomics with the available expertise at the VIB-KU Leuven Center for Brain & Disease Research, the team uncovered a microglial gene program, marked by strong expression of the gene SPP1. This was made possible by using NOVA-ST, a technique developed by the Stein Aerts lab (VIB-KU Leuven).
The findings of the VIB-KU Leuven research team pave the way for more refined Alzheimer’s therapies by defining the specific microglial program that drives effective plaque clearance.
“This opens doors to future therapies that may activate microglia without requiring antibodies. Understanding the importance of the Fc fragment helps guide the design of next-generation Alzheimer’s drugs,” concludes Prof. Bart De Strooper.
Reference: “The Alzheimer’s therapeutic Lecanemab attenuates Aβ pathology by inducing an amyloid-clearing program in microglia” by Giulia Albertini, Magdalena Zielonka, Marie-Lynn Cuypers, An Snellinx, Ciana Xu, Suresh Poovathingal, Marta Wojno, Kristofer Davie, Veerle van Lieshout, Katleen Craessaerts, Leen Wolfs, Emanuela Pasciuto, Tom Jaspers, Katrien Horré, Lurgarde Serneels, Mark Fiers, Maarten Dewilde and Bart De Strooper, 24 November 2025, Nature Neuroscience.
DOI: 10.1038/s41593-025-02125-8
The research team at the VIB-KU Leuven Center for Brain & Disease Research was financially supported by the European Research Council (ERC), Alzheimer’s Association USA, Research Foundation Flanders (FWO), Queen Elisabeth Medical Foundation for Neurosciences, Stichting Alzheimer Onderzoek – Fondation Recherche Alzheimer (STOPALZHEIMER.BE), KU Leuven, VIB, and UK Dementia Research Institute University College London.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.